Introduction
This is part 4 of the series of Calf Notes discussing amino acid nutrition in young calves. Previous Calf Notes on the topic (#238, #239, and #240) are available at Calf Notes.com. In this Calf Note, I’ll discuss the dynamics of amino acids in rumen microbial protein. This is a topic that has received scant attention for many years, so I’ll rely on some older data – including my M.S. thesis, published in 1983!
Change is the “name of the game”
Changing protein supply. Calves experience amazing changes, and in the case of dairy calves, in a very short time. We ask calves to transition from a monogastric method of digestion to a ruminant form of fermentation and digestion within a few weeks of life. During this time, calves often need to deal with transport, changes in housing, dehorning, and myriad other changes and stresses that may affect this important metabolic transition.
Let’s consider a week-old calf, drinking 6 liters of whole milk per day. At her age, she’s not eating any calf starter, because she’s consuming enough calories (about 4 megacalories of ME per day) to satisfy her appetite. The milk she’s drinking provides her with a well-balanced supply of amino acids that she can absorb and utilize for growth. We consider digestibility of whole milk to be about 97% and milk is mostly “true protein”, so she’s absorbing – and using – those amino acids for muscle growth. Note that although we think that milk is about 97% digestible, it may be less in this week old calf – see Calf Note #223 for more information. So, her intake of metabolizable protein (MP) will be about 194 grams per day. Nice. And, considering that milk generally by-passes the rumen, we can assume that 100% of her diet is “RUP” (rumen undegradable protein) at this young age.
Let’s fast forward 10 weeks. Our little calf isn’t so little anymore, and now she’s not drinking milk. Her diet consists of a 20% CP calf starter and ad libitum straw with about 5% CP. She’s eating about 2 kg of feed and about 5% of that is forage. So, her intake of protein will be about 385 grams per day. The interesting “bit” is that the protein (and amino acids) she eats are now subjected to ruminal fermentation, and most of the protein in her diet is degraded in the rumen by resident bacteria. It’s an amazing thing, but a young calf’s rumen can be just as active in fermenting carbohydrate and protein as an adult cow.
Between birth and “mature rumen function”, the calf transitions as the rumen grows in size and activity. One important consideration is the amount of dry feed that is the contribution of the rumen bacteria (and the amino acids in the bacteria) to the nutrition of the calf.
The “Bugs”
In adult ruminants, the protein leaving the intestine is a mixture of dietary protein that wasn’t degraded in the rumen (we call this rumen undegraded protein, or RUP), the protein in the microbial cells – mainly bacteria and protozoa that live in the rumen – this is called microbial crude protein, or MCP), and a smaller contribution of endogenous proteins from the animal. Most of the dietary protein degraded by the microbes in the rumen is used to make microbial protein. This process effectively changes the amino acid profile of what we feed the calf versus what the calf actually absorbs. In many cases, the amino acid profile of the microbes is better (more closely matches the needs of the calf) than the amino acids in the dietary protein, so this is a net “win” for the ruminant.
Ruminant nutritionists are familiar with the “RDP” and “RUP” approach to calculating the amino acid supply that the animal actually digests and absorbs. In the young calf, however, this change is unique and not well documented. So, we’ll consider some older data to determine how to calculate the amino acids that reach the calf’s small intestine.
Bacterial AA Composition. Considering all the changes occurring in the rumen during the transition from monogastric to ruminant, we might expect that the AA profile of the bacteria leaving the rumen might also change. Many studies have documented major changes in the types of rumen bacteria present in the rumen prior to and after weaning. Early in life, the rumen bacterial profile is quite different from that of a calf after weaning, which is mainly caused by changes in the rumen environment with increasing dry feed intake and fermentation in the rumen. In any event, the changes in the genera and species of rumen bacteria change profoundly during the first few months of life. These changes were first delineated using culture techniques by researchers such as Eadie (1962), Byrant et al. (1958) and Ziolecki and Briggs (1961). More recent studies, using genetic techniques (e.g., Baldwin et al., 2004; Li et al., 2012) have generally confirmed what we learned long ago, but with more depth in our understanding of the changes in types of bacteria predominating in the rumen.
As I mentioned previously, there are few data that document changing AA profiles in the rumen bacteria from calves during the ruminant transition. The only data readily available (from my personal library!) is my M.S. thesis from the University of New Hampshire in 1983. In that study, we evaluated the effect of weaning age (4 vs. 8 wk) and diet (pelleted vs. textured calf starter) on the flow of protein to the abomasum of the calf. During the study, we collected rumen fluid twice weekly from 2 to 11 wk of age and separated the bacteria by high-speed centrifugation. The cells were analyzed to determine the amount of protein and their amino acid profile. We also evaluated the changes in proportion of microbial vs. dietary protein in abomasal contents during this time.
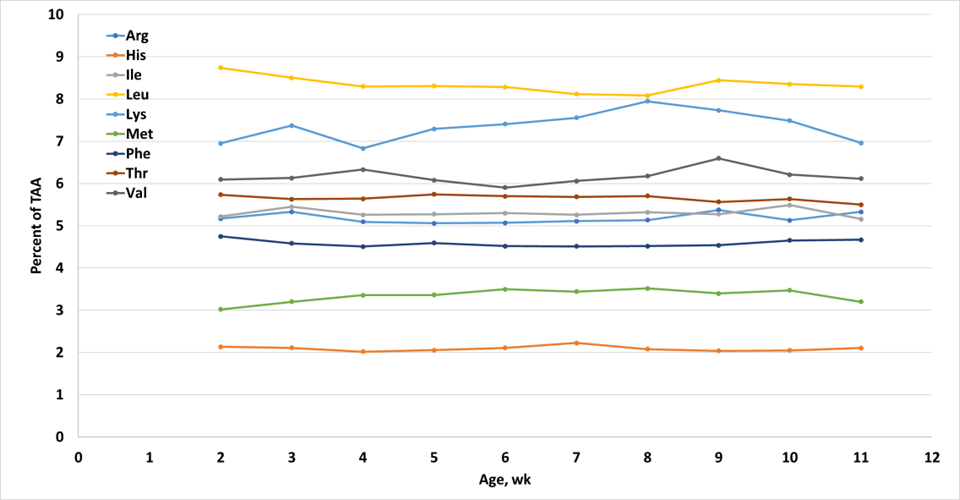
Weekly means of the four treatments used in the study were regressed on age to see if there were changes in AA profile over time. There were. Figure 1 and Table 1 show the differences that occurred over the 11 weeks of the study.
Of the essential amino acids, four AA changed with age – lysine, methionine, leucine, and phenylalanine (Table 1). However, as we can see in Figure 1, these changes were relatively small – e.g., lysine was about 7% of total AA in rumen bacteria of calves at 2 wk of age, and this increased to about 8% by 8 wk of age, but then again decreased to 7% at 11 wk of age. Similar changes occurred for the other AA. These changes were statistically significant, and it’s possible to model these changes with advancing age. However, the means presented are from only one study and these may not accurately represent the AA of bacteria in calves raised under different conditions. However, given the lack of other data on AA profile of rumen bacteria and the similarity of the values reported with those from adult cows (e.g., Sok et al., 2017), it seems reasonable to assume that the AA of rumen bacteria are (1) relatively similar to the AA in bacteria from adult cows and (2) don’t change dramatically with advancing age. This is important, as it allows us to more easily model AA flow of the young calf.
Table 1. Polynomial regression of age (weeks) on amino acid composition in rumen bacteria from Holstein calves fed milk replacer and calf starter. Adapted from Quigley, 1983.
| r2 | B0 | SE | P | B1 | SE | P | B2 | SE | P | |
| Arg | 0.210 | 5.390 | 0.260 | 0.001 | -0.095 | 0.090 | 0.300 | 0.008 | 0.007 | 0.240 |
| His | 0.050 | 2.100 | 0.120 | 0.001 | 0.000 | 0.040 | 0.950 | 0.000 | 0.003 | 0.950 |
| Ile | 0.070 | 5.250 | 0.150 | 0.001 | 0.020 | 0.051 | 0.700 | 0.000 | 0.004 | 0.700 |
| Leu | 0.570 | 9.120 | 0.190 | 0.001 | -0.249 | 0.065 | 0.001 | 0.016 | 0.005 | 0.010 |
| Lys | 0.420 | 6.170 | 0.439 | 0.001 | 0.386 | 0.151 | 0.010 | -0.026 | 0.011 | 0.030 |
| Met | 0.660 | 2.610 | 0.138 | 0.001 | 0.243 | 0.047 | 0.001 | -0.017 | 0.004 | 0.001 |
| Phe | 0.640 | 4.917 | 0.071 | 0.001 | -0.124 | 0.014 | 0.001 | 0.009 | 0.002 | 0.001 |
| Thr | 0.257 | 5.607 | 0.181 | 0.001 | 0.041 | 0.062 | 0.510 | -0.004 | 0.005 | 0.360 |
| Val | 0.090 | 6.118 | 0.422 | 0.001 | -0.003 | 0.145 | 0.980 | 0.001 | 0.011 | 0.900 |
Rumen Protozoa. In an adult ruminant, rumen protozoa contribute to the microbial protein reaching the intestine. Sok et al. (2017) pointed out that protozoa contribute about 16% of the microbial fraction and when we calculate the contribution of microbial AA to the total AA supply, it’s necessary to include the protozoal contribution. So, when it comes to calves, how do we calculate the contribution of protozoa?
For the first few weeks after birth, calves don’t maintain populations of protozoa (Pounden and Hibbs, 1950; Bryant and Small, 1958; Eadie et al., 1962). These become established later in life – the actual time frame depends on access to adult cattle and maintenance of a rumen pH greater than about 6.0 (Eadie, 1962; Fonty et al., 1988). Early in life, calves usually don’t have either access to adult cows or stable rumen pH, so we can assume that protozoa don’t contribute meaningfully to the AA profile of the microbial fraction. Figure 2 has an example of the differences in AA profile of bacteria and protozoa alone and a combination of protozoa and bacteria as suggested by Sok et al. (2017). We can see that protozoa have a different AA profile compared to rumen bacteria. Lysine looks particularly attractive, since protozoa have >10% of total AA as lysine whereas rumen bacteria contain less than 8% on average. However, when we consider the mixture of all types of bacteria and the smaller contribution of protozoa, the differences are diluted somewhat. When we compare the AA profile of the mixed rumen microbes from Sok et al. (2017) in Figure 2 (orange bars) with that of the mean AA values from young calves (black bars in Figure 2), we can see that there are some meaningful differences that should be accounted for when calculating the AA profile of the microbial fraction reaching the abomasum or intestine of the young calf. Finally,
It’s not clear exactly when calves will have a rumen environment stable enough to allow establishment of rumen protozoa, but on “rule of thumb” we might use is the intake of forage to help maintain rumen pH above the critical level (i.e., approximately 6.0). Most research suggests that forage helps stabilize the rumen pH and should be a reasonable benchmark for the time at which protozoa are available. Further, it’s not quite clear how long the rumen must “stabilize” prior to the establishment of a mature rumen protozoal population. Generally, a rule of thumb is that it requires about 14 days for meaningful changes to occur in the rumen after a change in diet; therefore, without any other guidance, we can assume that two weeks after calves are begin eating forage and can maintain a rumen pH >6.0, then the AA profile of microbial protein will change to reflect more the profile of Sok rather than that of Quigley.
Models of growth require us to predict nutrient requirements and nutrient supply (Van Amburgh et al., 2015; NASEM, 2021). The concept of changing microflora composition is an important consideration as we consider AA nutrition in young calves.
Summary
The AA profile of rumen microflora is dynamic in young calves. Although the AA profile of rumen bacteria in the young calf is similar to that of the bacteria in mature ruminants, changes occur in the proportion of certain AA as calves age. Also, the lack of ciliated protozoa in the rumen of young calves results in differences in AA profile of the mixed rumen flora that will change when calves can maintain a stable rumen environment. These changes should be incorporated into models to predict AA supply in calves from birth to four months of age. In future Calf Notes, we’ll apply these assumptions into a model to predict AA supply as calves transition from monogastric to functional ruminant.
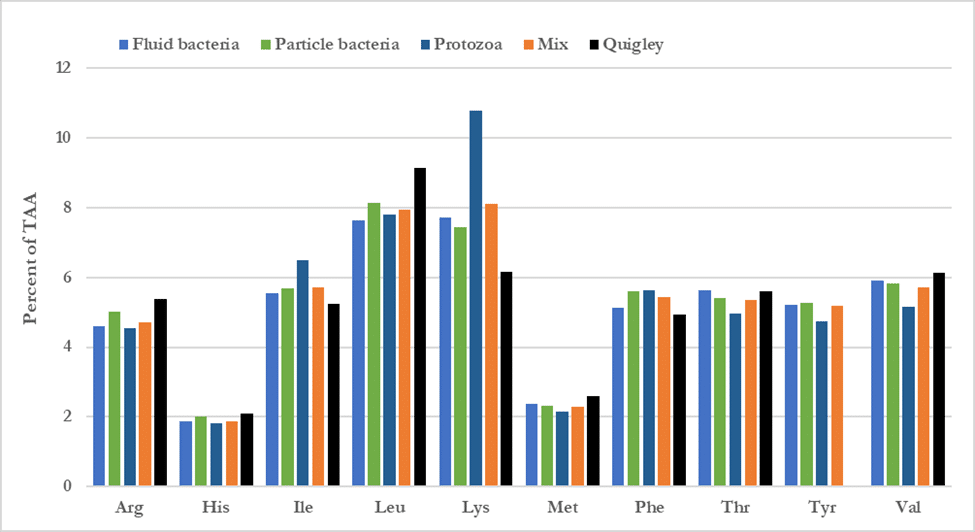
Figure 2. Amino acid profile of rumen bacteria, protozoa and a mixture of bacteria and protozoa typical of mature ruminants and mean AA data from young calves. Source: Quigley, 1983 and Sok et al., 2017.
References
Baldwin R. L. , K. R. McLeod, J. L. Klotz, and R. N. Heitmann. 2004. Rumen development, intestinal growth and hepatic metabolism in the pre- and postweaning ruminant. J. Dairy Sci. 87:55-65. https://doi.org/10.3168/jds.S0022-0302(04)70061-2.
Bryant, M. P., N. Small, C. Bouma, and I. Robinson. 1958. Studies on the composition of the ruminal flora and fauna of young calves. J. Dairy Sci., 41, 1747-1767. https://doi.org/10.3168/jds.S0022-0302(58)91160-3.
Eadie, J. M. 1962. The development of rumen microbial populations in lambs and calves under various conditions of management. J. Gen. Microbiol. 29:563-578. https://doi.org/10.1099/00221287-29-4-563.
Eadie, J. M. 1962. Inter-relationships between certain rumen ciliate protozoa. J. Gen. Microbiol. 29:579-588. https://doi.org/10.1099/00221287-29-4-579.
Fonty, G., J. Senaud, J. P. Jouany, and P. Gouet. 1988. Establishment of ciliate protozoa in the rumen of conventional and conventionalized lambs: influence of diet and management conditions. Can. J. Microbiol. 34:235–41. https://doi.org/10.1139/m88-044.
Godfrey, N. W. 1961. The functional development of the calf. II. Development of rumen function in the calf. J. Agric. Sci. 57:177. https://doi.org/10.1017/S0021859600047651.
Li, R. W., E. E. Connor, C. Lee, R. L. Baldwin, and M. E. Sparks. 2012. Characterization of the rumen microbiota of pre-ruminant calves using metagenomic tools. Environmental Microbiology. 14:129-139. https://doi.org/10.1111/j.1462-2920.2011.02543.x.
NASEM. 2021. Nutrient Requirements of Dairy Cattle. 8th Revised Ed. Washington, DC. The National Academies Press. https://doi.org/10.17226/25806.
Pounden, W. D., and J. W. Hibbs. 1950. The development of calves raised without protozoa and certain other characteristic rumen microorganisms. J. Dairy Sci. 33:639-644. https://doi.org/10.3168/jds.S0022-0302(50)91948-5.
Van Amburgh, M. E., E. A. Collao-Saenz, J. Higgs, D. A. Ross, E. B. Recktenwald, E. Raffrenato, L. E. Chase, T. R. Overton, J. K. Mills, and A. Foskolos. 2015. The Cornell Net Carbohydrate and Protein System: Updates to the model and evaluation of version 6.5. J. Dairy Sci. 98:6361-6380. http://dx.doi.org/10.3168/jds.2015-9378.
Quigley, J. D. 1983. The effect of weaning age and ration on the development of microbial protein production in young calves. MS Thesis. University of New Hampshire, Durham.
Quigley, J. D., C. G. Schwab, and W. E. Hylton. 1985. Development of rumen function in calves: Nature of protein leaving the abomasum. J. Dairy Sci. 68:694-702. https://doi.org/10.3168/jds.S0022-0302(85)80875-4.
Sok, M., D. R. Ouellet, J. L. Firkins, D. Pellerin, and H. Lapierre. 2017. Amino acid composition of rumen bacteria and protozoa in cattle. J. Dairy Sci. 100:5241–5249. https://doi.org/10.3168/jds.2016-12447.
Ziolecki, A., and C.A.E. Briggs. 1961. The microflora of the rumen of the young calf. II. Source, nature and development. J. Appl. Bacteriol. 24:148. https://doi.org/10.1111/j.1365-2672.1961.tb00247.x.