Introduction
Apparent efficiency of absorption (AEA) has been used for many years as a practical way to describe the success of colostral IgG transfer in the newborn calf. The calculation is straightforward: estimate the amount of IgG apparently present in the calf after feeding, divide that by the amount of IgG fed, and express the result as a percentage. The value is useful because it gives us a way to compare calves, compare feeding programs, and evaluate management. However, the word apparent is important.
Let’s begin with a little context. We use AEA to predict serum IgG concentration in a calf at a certain time after birth:
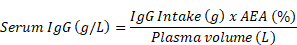
The IgG intake is colostrum IgG concentration × colostrum intake, and plasma volume is usually calculated as body weight (kg) × a factor to estimate plasma volume, often 9%.
We can rearrange the equation to calculate AEA:
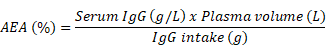
The AEA is an estimate of how efficient a calf is in absorbing IgG from the colostrum. Some calves, of course, will absorb more IgG per unit of ingested IgG, and some less. So, it’s an important measure of colostrum management on the farm. Factors such as prepartum heat stress (Tao et al., 2012; Dado-Senn et al., 2020), pasteurization of colostrum (Elizondo-Salazar and Heinrichs, 2009; Robbers et al., 2021), and dystocia (Waldner et al., 2009; Murray et al. 2015) and other factors have been reported to affect AEA.
However, AEA is not a direct measurement of how much IgG crossed the intestinal wall. It is an estimate based on the IgG concentration measured in serum or plasma, combined with an estimate of plasma volume. That means the value is influenced not only by intestinal absorption, but also by how IgG is distributed in body fluids after it is absorbed. Thus, AEA is not simply a measure of intestinal absorption. It is also influenced by how IgG distributes within the calf after it is absorbed. This distinction may help explain why AEA is never 100%, why it varies so much among calves, and why some of that variation may reflect physiology beyond intestinal uptake alone.
The Basic Concept
When a calf consumes colostrum, immunoglobulin G (IgG) moves from the intestinal tract into the body via the process of pinocytosis. But once absorbed, IgG does not remain only in the bloodstream. Instead, it distributes between at least two major fluid compartments:
- the vascular space, meaning the plasma within blood vessels
- the extravascular space, including interstitial fluids outside the blood vessels
If we measure serum IgG at one point in time, we are measuring only the concentration of IgG in the vascular compartment at that moment. We are not measuring total IgG absorbed into the body – we are measuring how much IgG is present in the vascular space at a specific moment in time.
This is important because serum concentration depends on both:
- how much IgG has been absorbed, and
- how much fluid volume that IgG is distributed into
Thus, two calves may absorb similar amounts of IgG, but if IgG distributes differently between vascular and extravascular spaces, the measured serum concentration may differ.
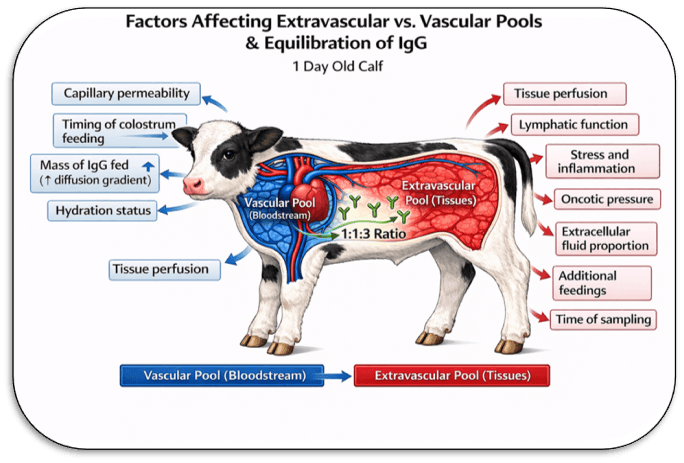
“Extravascular Space”
Physiologically, body fluids are typically described as intracellular and extracellular compartments. The extracellular fluid includes both plasma (within blood vessels) and interstitial fluid (outside blood vessels). The intracellular compartment represents fluid within the cells of the body. These compartments are estimated using specific markers.
Estimates of total body water in calves range from about 72% to 88% (Thornton and English, 1975) based on research published from 1920 to 1975. Most of the water in calves is located in the extracellular space—80% or more of total body water has been reported to be extracellular (Wagstaff et al., 1992). However, only a small portion of this fluid is contained within the vascular space where serum IgG is measured.
When considering IgG absorption, we typically measure plasma volume (PV), which is part of the extracellular compartment. Plasma volume in calves is usually measured using a type of marker such as Evans Blue dye (Thornton and English, 1978; Wagstaff et al., 1992; Quigley et al., 1998; Cabral et al., 2015) or radioisotopes such as ¹³¹I-labelled serum albumin (Möllerberg et al., 1975; Wagstaff et al., 1992). Plasma volume increases from birth to 90 days of age—Möllerberg et al. (1975) reported that PV increased from 5.3% at birth to 6.5% at 1 day of age and 4.9% at 90 days. They also reported that blood volume values were 8.4%, 9.3% and 7.0% of BW, respectively.
Plasma volume values reported by Möllerberg et al. (1975) are lower than values commonly reported when Evans Blue dye is used. For example, Quigley et al. (1998) reported PV of approximately 9% of BW, and Cabral et al. (2015) reported similar values. Interestingly, van Waversveld and Bruchem (1985) concluded that Evans Blue may estimate blood volume rather than true plasma volume.
For the purposes of understanding IgG absorption, it is useful to think of two functional compartments: the vascular space, where we measure IgG, and the extravascular space, where IgG may also distribute after absorption.
Let’s use an example of a 40-kg calf at 1 day of age to calculate body water compartments and consider the dynamic nature of serum IgG measurements. In this example, we will assume that total body water is 75% of body weight, that 80% of total body water is in the extracellular compartment, and that plasma volume is 9% of body weight.
- Body weight (BW) = 40 kg
- Total body water (TBW) = 40 × 0.75 = 30 kg
- Extracellular fluid = 30 × 0.80 = 24 kg
- Intracellular fluid = 30 − 24 = 6 kg
- Plasma volume (intravascular; part of extracellular fluid) = 40 × 0.09 = 3.6 kg
- Interstitial fluid (extravascular; part of extracellular fluid) = 24 − 3.6 = 20.4 kg
Thus, in this example, the calf has approximately 24 kg of extracellular fluid, but only 3.6 kg is within the vascular space where serum IgG is measured. The ratio of interstitial to plasma fluid is about 5.7:1; in other words, the interstitial fluid compartment is nearly six times larger than the plasma compartment.
Volume of Distribution
In pharmacology, the distribution of a substance between vascular and extravascular compartments is often described using the concept of volume of distribution (Vd). This value reflects the extent to which a compound moves out of the bloodstream and into surrounding tissues and fluids. Importantly, Vd does not necessarily correspond to a physical or anatomical volume. Instead, it represents the apparent volume into which a substance has distributed at the time it is measured.
This concept may be useful when considering IgG in the newborn calf. Although the total extravascular fluid space is several times larger than plasma volume, IgG does not appear to distribute uniformly or instantaneously into this entire space. Instead, the observed distribution of IgG between vascular and extravascular compartments is much smaller, where the ratio is approximately 1.3:1. This is much smaller than the anatomical ratio of interstitial to plasma fluid, indicating that IgG has equilibrated with only a portion of the total extravascular space at the time of measurement.
IgG distributes into extravascular fluids, including interstitial space, and this has been demonstrated in multiple species using lymph and tissue fluid measurements. Thus, serum IgG concentration reflects the balance between absorption and distribution, not absorption alone.
Movement of IgG from the vascular compartment into extravascular fluids occurs primarily through normal capillary exchange processes and fluid movement, rather than through specific receptor-mediated transport. The neonatal Fc receptor (FcRn), which plays a key role in intestinal absorption of IgG, is also involved in protecting IgG from degradation and recycling it within the body. However, its primary function appears to be maintenance of IgG within the circulation rather than driving its distribution into extravascular spaces.
Why AEA Will Never Be 100%
Under field conditions, and probably even under ideal experimental conditions, AEA should not be expected to reach 100%. There are several reasons for this.
First, not all IgG fed in colostrum is absorbed. Intestinal absorption is inherently incomplete. Gut closure begins soon after birth, and even in the first hours of life transport is limited by time, receptor activity, competition among proteins, and normal biological variation.
Second, AEA is based on serum or plasma IgG concentration, not total body IgG. If some IgG has moved into extravascular fluids by the time blood is sampled, then the measured value in plasma will underestimate the total amount of IgG absorbed.
Third, IgG distribution is dynamic. As IgG moves between vascular and extravascular compartments, the measured serum concentration changes over time, even if total IgG in the body does not. Thus, AEA reflects not only how much IgG was absorbed, but also where that IgG is at the time it is measured.
A Dynamic System, Not a Static One
One of the easiest mistakes is to think of IgG absorption as a simple event: colostrum is fed, IgG crosses the intestine, and serum IgG rises to a final value. In reality, this is a dynamic process.
Several events are occurring at the same time:
- IgG is being delivered to the small intestine
- IgG is being absorbed across the gut wall
- IgG is entering the vascular space
- IgG is distributing into extravascular fluids
- fluid shifts are occurring in the newborn calf
- the calf may be urinating, standing, becoming more active, and adapting to extrauterine life
Therefore, the measured serum IgG concentration at any single sampling time reflects the balance of these ongoing processes. It is not merely a measure of “absorption.” It is a snapshot taken during a moving physiological process.
Vascular and Extravascular Space
The two-compartment idea is conceptually simple and may be helpful for understanding colostral IgG. The two-compartment concept provides a simple way to think about IgG dynamics. Immediately after absorption, IgG enters the bloodstream. From there, it does not remain confined to plasma. Over time, it equilibrates with extravascular fluids. The rate and extent of this equilibration are likely influenced by several factors, including age, time after feeding, hydration status, capillary permeability, health status, and environmental conditions.
At present, these factors are not well accounted for in routine AEA calculations. Yet they may contribute substantially to the variation observed among calves. This does not make AEA useless. On the contrary, AEA remains a valuable practical index. But it does suggest that part of the unexplained variation in AEA may be due to post-absorptive distribution rather than absorption alone.
Summary
Our estimates of AEA (and serum IgG) are estimates at best. They represent a “snapshot” point in time in a dynamic system of equilibration that occurs after the calf is fed colostrum. Understanding the events affecting factors in the AEA equation – particularly plasma volume – will assist us in improving predictions of AEA and how calves absorb – and metabolize – IgG.
References
Cabral, R. G., C. E. Chapman, E. J. Kent, and P. S. Erickson. 2015. Estimating plasma volume in neonatal Holstein calves fed one or two feedings of a lacteal-based colostrum replacer using Evans blue dye and hematocrit values at various time points. Can. J. Anim. Sci. 95:293-298 doi:10.4141/CJAS-2014-176 293.
Dado-Senn, B., L. Vega Acosta, M. Torres Rivera, S. L. Field, M. G. Marrero, B. D. Davidson, S. Tao, T. F. Fabris, G. Ortiz-Colón, G. E. Dahl, and J. Laporta. Pre- and postnatal heat stress abatement affects dairy calf thermoregulation and performance. J. Dairy Sci. 103:4822-4837. https://doi.org/10.3168/jds.2019-17926.
Elizondo-Salazar, J. A., and A. J. Heinrichs. 2009. Feeding heat-treated colostrum or unheated colostrum with two different bacterial concentrations to neonatal dairy calves. J. Dairy Sci. 92 :4565–4571 doi:10.3168/jds.2009-2188.
Möllerberg, L., L. Ekman and S. Jacobsson. 1975. Plasma and blood volume in the calf from birth till 90 days of age. Acta Vet. Scand. 16:178-185.
Murray, C. F., D. M. Veira, A. L. Nadalin, D. M. Haines, M. L. Jackson, D. L. Pearl, and K. E Leslie. 2015. The effect of dystocia on physiological and behavioral characteristics related to vitality and passive transfer of immunoglobulins in newborn Holstein calves. Can. J. Vet. Res. 79:109–119.
Quigley, J. D. III, J. J. Drewry, and K. R. Martin. 1998. Estimation of plasma volume in Holstein and Jersey calves. J. Dairy Sci. 81:1308-1312.
Robbers, L., R. Jorritsma, M. Nielen, and A. Koets. 2021. A scoping review of on-farm colostrum management practices for optimal transfer of immunity in dairy calves. Front. Vet. Sci., 18 July 2021. 8-2021. https://doi.org/10.3389/fvets.2021.668639.
Tao, S., A. P. A. Monteiro, I. M. Thompson, M. J. Hayen, and G. E. Dahl. 2012. Effect of late-gestation maternal heat stress on growth and immune function of dairy calves. J. Dairy Sci. 95:7128–7136. http://dx.doi.org/ 10.3168/jds.2012-5697.
Thornton, J. R., and P. B. English. 1978. Body water of calves: Change in distribution with diarrhoea. Br. Vet. J. 134:445-453.
Wagstaff, A. J., I. Maclean, A. R. Michell, and P. H. Holmes. 1992. Plasma and extracellular volume in calves: comparison between isotopic and ‘cold’ techniques. Res. Vet. Sci., 53:271-273.
Waldner, C. L., and L. B. Rosengren . 2009. Factors associated with serum immunoglobulin levels in beef calves from Alberta and Saskatchewan and association between passive transfer and health outcomes. Can Vet J. 50:275–281.