Introduction
Fat-soluble vitamins are essential to young calves and should be included in both milk replacers and calf starters. The primary fat-soluble vitamins – A, D, and E – are critical to calf health and growth and should be included in milk replacers, starters, and growers.
I conducted a review of the scientific literature regarding vitamins A, D, and E and reviewed the recommendations from the 2021 NASEM committee in the “Nutrient Requirements for Dairy Cattle” (NASEM, 2021). To evaluate the NASEM recommendations, I utilized several calf growth models and determined requirements based on liquid and dry feed intakes at various ages, using typical feeding programs. I evaluated minimum requirements for each vitamin based on intake from both milk replacer and calf starter to always meet or exceed the NASSEM recommendation. For more information on this approach, see Appendix B in Calf Note #243. Here is a summary of the literature and my recommendations (“Adjusted”) when they differed from those of NASEM.
Vitamin E. The NASEM Committee recommended added vitamin E concentration of 2 IU/kg BW, or approximately 125 IU/d (Table 1). They also stated that this rate of supplementation may be inadequate for rapidly growing calves. It has been shown that rate of BW gain affects vitamin E requirement (NASEM, 2021). Thus, when high-growth programs are implemented (i.e., programs that allow >700 g ADG), additional vitamin E equivalent should be included.
Recommended vitamin E requirements in calf diets have increased as researchers reported the important role of the vitamin in calf immune response (e.g., Reddy et al., 1987a,b). Vitamin E has been studied in greater detail than other vitamins since publication of the 2001 Dairy NRC. Much of the work has documented a role for α-tocopherol in promoting immune response, mainly by providing antioxidant capacity to the animal and reducing effects of oxidative stress on immune competence (Abuelo et al., 2019). Effects of supplemental vitamin E on indices of immune response (lymphocyte numbers, lymphocyte activation, antibody titer in response to vaccination) have been reported in Holstein calves and Japanese Wagyu calves (Otomaru et al., 2013; 2015). Vitamin E may play a role in maturation of the immune system as indicated by production of nitric oxide by blood mononuclear leukocytes (Rajaraman et al., 1998). Production of nitric oxide in juvenile leukocytes is greater than that of mature cells; vitamin E may play a role in maturation of these cells to ameliorate potential cell damage caused by excess NO production. Conversely, Schmidt et al. (1998) reported no effect of vitamin E supplementation (188 vs. 354 IU/d) on hematology of calves vaccinated with Shiga toxin at five and eight weeks of age. Also, Waldner et al. (2017) reported that calves with serum vitamin E less than adequate for their age (2–7 d old, <0.8 μg/mL; >7 d old, <0.5 μg/mL) were 3.2 times more likely to be treated for enteritis than calves with higher concentrations.
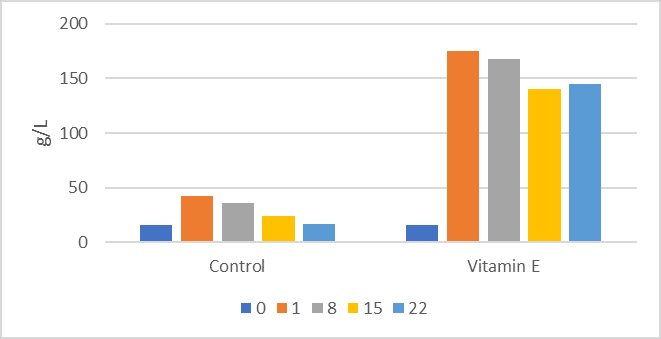
Vitamin E status of neonatal calves depends to a large extent on the dry cow diet and consumption of colostrum, which is the primary route for delivery of fat-soluble vitamins in newborn calves (Quigley and Bernard, 1995). Supplementation of colostrum increased vitamin E status of newborn calves (Quigley and Bernard, 1995). It is clear that variability in fat content in general and fat-soluble vitamin content in colostrum may affect vitamin E and antioxidant status of neonates. Mohri et al. (2005) reported that injection of vitamin E (300 U α-tocopherol acetate/45 kg BW) and selenium (6 mg/45 kg BW) altered hematology of young calves, generally improving numbers of lymphocytes and indices of antioxidant capacity. Vaccination of newborn Jersey calves with 2,000 IU vitamin E once weekly for the first two weeks of life vastly improved serum IgG concentrations (Figure 1; Pekmezci and Cakiroglu, 2009). Serum IgG concentrations in excess of 100 g/L early in life suggest dramatic increase in onset of active immunity and should be considered supraphysiological.
Reddy et al. (1987b) recommended 125 IU/d as the optimal level of supplementation for young Holstein calves. However, liquid feeding rates in this study were low (i.e., 8% of BW; Reddy et al., 1987a) and it has been shown that vitamin E requirement increases with increasing growth (Nonnecke et al., 2010). Therefore, supplementation with vitamin E or vitamin E equivalents may need to vary depending on CMR feeding rate and projected growth rte.
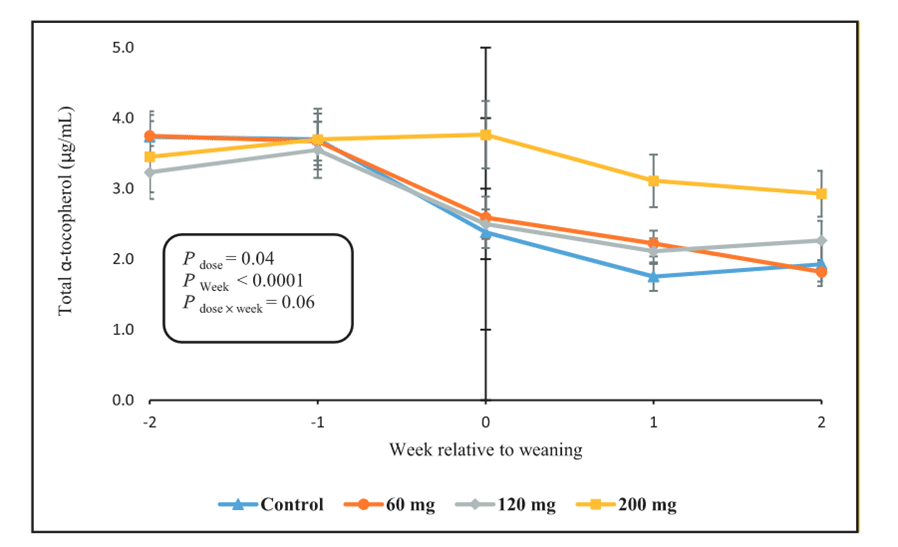
Weaning is an important transition in the life of young calves and reduced antioxidant status of weaned calves (Majlesi et al., 2021) may have deleterious effects on post-weaning resistance to disease. Recently, Lashkari et al. (2022) reported that plasma RRR-α-tocopherol was 0.8 µg/ml at weaning (61 d of age). Generally, plasma vitamin E concentrations less than 0.5 to 1.0 µg/ml are considered inadequate in most species (McDowell et al., 1996) and plasma vitamin E of 0.6 to 1.6 µg/ml may result in muscular dystrophy in calves (Adams, 1982). Decline in plasma vitamin E at weaning may be related to transition from a diet high in fat (from milk or milk replacer) to one typically containing <5% fat on a DM basis. Further, the nature of the fat (potentially unsaturated) may interfere with α-tocopherol absorption by providing inadequate substrate for micellarization during digestion and intestinal transport. Postweaning malabsorption of α-tocopherol has been reported (Reddy et al., 1987a). Feeding 60 or 120 mg/kg of RRR-α-tocopherol was insufficient to maintain plasma vitamin E concentrations, whereas 200 mg/kg maintained plasma levels for 2 weeks after weaning (Lashkari et al., 2022; Figure 2). These authors recommended that α-tocopherol concentrations in diets fed to calves around weaning should be 200 mg/kg of DM. These authors also compared sources of vitamin E – 200 mg/d of RRR-α-tocopherol (ALC), 200 mg/d of RRR-α-tocopheryl acetate (ACT), or 200 mg/d of all-rac-α-tocopheryl acetate (SYN). Concentrations of plasma α-tocopherol at weaning were 2.7, 2.1, 1.1, and 0.8 µg/ml for ALC, ACT, SYN, and Control, respectively (Lashkari et al., 2022). The authors concluded that 200 mg/kg of ALC was the most effective inclusion to maintain plasma vitamin E levels around weaning.
Vitamin A. The new NASEM recommendation for intake of vitamin A is 110 IU per kg of BW (Table 1) or 11,000 IU/kg in milk replacer and 3,700 IU/kg in calf starter and grower (Table 2).
Kume and Toharmat (2001) reported results with 46 Holstein calves at birth and 6 d of age. Concentrations of both β-carotene and plasma vitamin A increased from birth to 6 d. Plasma β-carotene and plasma vitamin A were positively correlated with fecal DM, but no correlations were observed between plasma vitamins and erythropoiesis components. These results suggest that β-carotene status of calves at 6 days of age is dependent on colostrum concentrations of β-carotene and affects the occurrence of diarrhea, and vitamin A status depends on colostral vitamin A and placental vitamin A transfer during gestation. Eicher et al. (1994) reported that combination of vitamins A and E may improve function of leukocytes from young calves to 6 wk of age. In terms of manufacturing, emulsification of fat-soluble vitamins in general, and vitamin A in particular, improve absorption (Jacobson et al., 1954). Waldner et al. (2017) reported that calves with serum vitamin A less than 0.14 μg/mL were 2.8 times more likely to die, indicating the role that vitamin A plays in overall immune response.
Vitamin D. The new NASEM recommendation for intake of vitamin D is 32 IU per kg of BW (Table 1) or 3,200 IU/kg in milk replacer and 1,100 IU/kg in calf starter and grower (Table 2).
The vitamin D status of animals is reliably indicated by the concentration of the 25-hydroxyvitamin D (25(OH)D) metabolite in serum or plasma, with a concentration of 30 ng/mL proposed as a lower threshold for sufficiency. Calves are often born with vitamin D insufficiency (< 8 ng/ml of 25(OH)D) and may remain in this state for as long as 3 months (Flores-Villalva et a. 1021). As summarized by Nelson et al. (2016), serum 25(OH)D concentrations are approximately 15 ng/mL at birth and remained ≤15 ng/mL through 1 mo of age if fed pasteurized waste milk with little to no summer sun exposure. In contrast, serum 25(OH)D of calves fed milk replacer containing 6,600 and 11,000 IU of vitamin D3/kg of dry matter were 59 ± 8 and 98 ± 33 ng/mL, respectively, at 1 mo of age (Nelson et al., 2019). Calves similarly supplemented indicated that serum 25(OH)D achieved at approximately 1 mo of age would increase 6 to 7 ng/mL for every 1,000 IU of vitamin D3/kg of dry matter of milk replacer. Blakely et al. (2019) suggested that supplementation of pasteurized waste milk with vitamins A, D, and E (0.25 or 0.50 ml/day of a product containing 50,000 IU of vitamin A as retinyl-palmitate, 50,000 IU of vitamin D3 and 500 IU of vitamin E as RRR-α-tocopherol per milliliter of product) plus 0.5 ml of the same supplement injected at birth resulted in lower growth rates to 28 d of age than unsupplemented calves, probably due to excess vitamin supplementation. Thus, there appears to be an upper limit to supplementation with fat-soluble vitamins. Finally, researchers in China suggested that feeding 25(OH)D in CMR consumed more dry feed and grew faster than non-supplemented controls (but similarly to calves fed vitamin D3) and reduced indices of stress associated with weaning at 60 d compared to unsupplemented controls (Xu et al., 2021; Wang et al., 2022).
Summary and recommendations. Requirements for fat-soluble vitamins (IU/d) are in Table 1. These equations may be incorporated into predictive models. We recommend Adjusted requirements in preference to those published by NASEM. We recommend increasing Vitamin A and D concentrations by 10% to account for normal oxidation in feeds during manufacturing and storage of feeds. Table 5 contains recommended inclusions for fat soluble vitamins in milk replacer and starter to provide adequate intake for calves from 35 to 125 kg.
Table 1. Fat-soluble vitamin minimum recommended intake, IU/day, NASEM, 2021 and Adjusted.
| NASEM | Adjusted | ||
| Vitamin | IU/day | IU/day (min) | |
| Vit A | 110 * BW | 110 * BW * 1.1 | |
| Vit D | 32 * BW | 32 * BW * 1.1 | |
| Vit E | 2 * BW | 2 * BW | |
| Vit E equivalent* | 4 * BW |
Table 2. NASEM and Adjusted recommended concentrations of fat soluble vitamins in milk replacer and starter to provide adequate intake for calves from 35 to 125 kg. Values are on a DM basis. Adjusted calculations based on non-linear minimization of vitamin intake above requitements and modeling of daily intake vs. requirement (IU/d).
| NASEM, IU/kg DM | Adjusted, IU/kg DM | ||||||
| Vitamin | CMR | Starter | Grower | CMR | Starter | Grower | |
| Vit A | 11,000 | 3,700 | 3,700 | 11,000 | 4,500 | 4,500 | |
| Vit D | 3,200 | 1,100 | 1,100 | 3,500 | 1,300 | 1,300 | |
| Vit E | 200 | 67 | 67 | 200 | 80 | 80 | |
| Vit E equiv.* | 300 | 160 | 160 | ||||
References
Abuelo, A., J. Hernández, J. L. Benedito, and C. Castillo. 2019. Redox biology in transition periods of dairy cattle: Role in the health of periparturient and neonatal animals. Antioxidants (Basel). 13:20. https://doi.org/10.3390/antiox8010020.
Adams, C. 1982. Feedlot cattle need supplemental vitamin E. Feedstuffs. 54:24.
Blakely, L. P., M. B. Poindexter, R. L. Stuart, and C. D. Nelson. 2019. Supplementing pasteurized waste-milk with vitamins A, D, and E improves vitamin status of dairy calves. The Bovine Practitioner. 53:134–141. https://doi.org/10.21423/bovine-vol53no2p134-141.
Flores-Villalva, S., M. B. O’Brien, S. Reid, S. Lacey, S. V. Gordon, C. Nelson, and K. G. Meade. 2021. Low serum vitamin D concentrations in spring-born dairy calves are associated with elevated peripheral leukocytes. Sci. Rep. 11:18969. https://doi.org/10.1038/s41598-021-98343-8.
Jacobson, N. L., R. S. Allen, J. T. Blake, and P. G. Homeyer. 1954. The effect of method of administration on the absorption and storage of vitamin A by dairy calves: Two figures. J. Nutr. 54:143–153. https://doi.org/10.1093/jn/54.1.143.
Kume, S., and T. Toharmat. 2001. Effect of colostral β-carotene and vitamin A on vitamin and health status of newborn calves. Livestock Prod. Sci. 68:61-65. https://doi.org/10.1016/S0301-6226(00)00214-1.
Lashkari, S., S. K. Jensen, and M. Vestergaard. 2022. Response to different sources of vitamin E orally injected and to various doses of vitamin E in calf starter on the plasma vitamin E level in calves around weaning. Animal. 16:100492. https://doi.org/10.1016/j.animal.2022.100492.
Majlesi, A., S. P. Yasini, S. Azimpour, and P. Mottaghian. 2021. Evaluation of oxidative and antioxidant status in dairy calves before and after weaning. Bulg. J. Vet. Med. 24:184-190. https://doi.org/10.15547/bjvm.2270.
McDowell, L. R., S. N. Williams, N. Hidiroglou, C. A. Njeru, G. M. Hill, L. Ochoa, and N. S. Wilkinson. 1996. Vitamin E supplementation for the ruminant. Anim. Feed Sci. Technol. 60:273-296. https://doi.org/10.1016/0377-8401(96)00982-0.
Mohri, M. H., A. Seifi, and J. Khodadadi. 2005. Effects of preweaning parenteral supplementation of vitamin E and selenium on hematology, serum proteins, and weight gain in dairy calves. Comp. Clin. Pathol. 14: 149–154. https://doi.org/10.1007/s00580-005-0581-3.
National Academies of Sciences, Engineering, and Medicine. 2016. Nutrient Requirements of Beef Cattle: Eighth Revised Edition. Washington, DC: The National Academies Press. https://doi.org/10.17226/19014.
Nelson, C. D., J. D. Lippolis, T. A. Reinhardt, R. E. Sacco, J. L. Powell, M. E. Drewnoski, M. O’Neil, D. C. Beitz, and W. P. Weiss. 2016. Vitamin D status of dairy cattle: Outcomes of current practices in the dairy industry. J. Dairy Sci. 99:10150-10160. https://doi.org/10.3168/jds.2016-11727.
Nonnecke, B. J., M. R. Foote, B. L. Miller, D. C. Beitz, and R. L. Horst. 2010. Short communication: Fat-soluble vitamin and mineral status of milk. replacer-fed dairy calves: Effect of growth rate during the preruminant period. J. Dairy Sci. 93:2684–2690. https://doi.org/10.3168/jds.2009-2892.
Otomaru, K. S. Saito, K. Endo, M. Kohiruimaki, S. Fukuyama, and H. Ohtsuka. 2013. Effect of supplemental vitamin E on antibody titer in Japanese black calves vaccinated against bovine herpesvirus-1. J. Vet. Med. Sci. 75:1671–1673. https://doi.org/10.1292/jvms.13-0215.
Otomaru, K., S. Saito, K. Endo, M. Kohiruimaki, and H. Ohtsuka. 2015. Effect of supplemental vitamin E on the peripheral blood leukocyte population in Japanese Black calves. J. Vet. Med. Sci. 77:985–988. https://doi.org/10.1292/jvms.15-0060.
Pekmezci, D., and D. Cakiroglu. 2009. Investigation of immunomodulatory effects of levamisole and vitamin E on immunity and some blood parameters in newborn Jersey calves. Vet. Res. Commun. 33:711–721. https://doi.org/10.1007/s11259-009-9220-9.
Quigley III, J. D., and J. K. Bernard. 1995. Effects of addition of vitamin E to colostrum on serum α‐tocopherol and immunoglobulin concentrations in neonatal calves. Food and Ag. Immunol. 7:295-298. https://doi.org/10.1080/09540109509354887.
Rajaraman, V., B. J. Nonnecke, S. T. Franklin, D. C. Hammel, and R. L. Horst. 1998. Effects of vitamin A and E on nitric oxide production by blood mononuclear leukocytes from neonatal calves fed milk replacer. J. Dairy Sci. 81:3278–3285. https://doi.org/10.3168/jds.S0022-0302(98)75892-8.
Reddy, P. G., J. L. Morrill, and R. A. Frey. 1987a. Vitamin E requirements of dairy calves. J. Dairy Sci. 70:123-129. https://doi.org/10.3168/jds.S0022-0302(87)79987-1.
Reddy, P. G., J. L. Morrill, H. C. Minocha, and J. S. Stevenson. 1987b. Vitamin E is immunostimulatory in calves. J. Dairy Sci. 70:993-999. https://doi.org/10.3168/jds.S0022-0302(87)80104-2.
Schmidt, N., T. Luhmann, L. Hüther, U. Meyer, S. A. Barth, L. Geue, C. Menge, J. Frahm, and S. Dänicke. 2018. Effect of vitamin E supplementation in milk replacer and Shiga toxoid vaccination on serum α-tocopherol, performance, haematology and blood chemistry in male Holstein calves. Anim. Physiol. Anim. Nutr. 102:1167-1180. https://doi.org/10.1111/jpn.12926.
Waldner, C. L. and F. D. Uehlinger. 2016. Factors associated with serum vitamin A and vitamin E concentrations in beef calves from Alberta and Saskatchewan and the relationship between vitamin concentrations and calf health outcomes. Can. J. Anim. Sci. 97:65-82. https://doi.org/10.1139/cjas-2016-0055.
Wang, L. H., C. R. Zhang, Q. Y. Zhang, H. J. Xu, G. Z. Feng, G. N. Zhang, and Y. G. Zhang. 2022. Effects of feeding different doses of 25-hydroxyvitamin D3 on the growth performance, blood minerals, antioxidant status and immunoglobulin of preweaning calves. Anim. Feed Sci. Tech. 285:115220. https://doi.org/10.1016/j.anifeedsci.2022.115220.
Xu, H. J., L. H. Wang, Q. Y. Zhang, X. Jiang, C. R. Zhang, and Y. G. Zhang. 2021. Effects of 25-hydroxyvitamin D3 on growth performance, fecal scores, vitamin D3 metabolites, antioxidant status, and inflammatory and stress-related parameters in weaning calves. Anim. Feed Sci. Tech. 281:114946. https://doi.org/10.1016/j.anifeedsci.2021.114946.